TRANSFORMING
HEALTHCARE
RESEARCH IN AFRICA
TRANSFORMING HEALTHCARE RESEARCH IN AFRICA
AI-DRIVEN TRANSFORMATION IN CLINICAL TRIALS
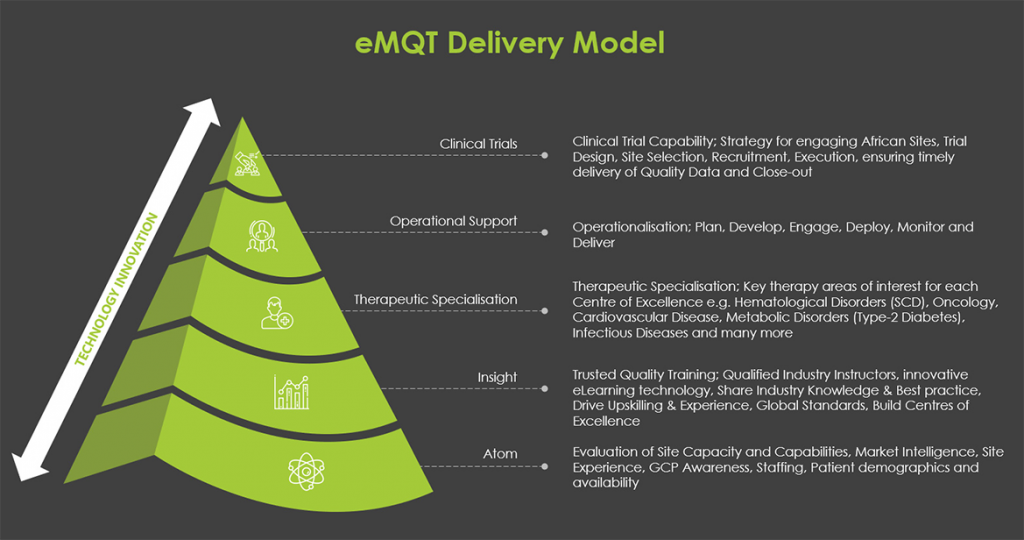
Emerging Markets Quality Trials
Welcome to eMQT where we enable two clear solutions for the pharmaceutical industry:
We stand as a powerhouse where AFRICAN LIVES MATTER! Serving as the unyielding bridge between the pharmaceutical industry and Africa, we leverage the expertise of research healthcare professionals and ground-breaking technological innovation. Our mission is to not only grant access to patients but also to generate high-quality, evidence-based patient data, fuelled by the driving forces of training and education.
We leverage technological innovation to redefine global clinical research. Our transformative, technology-driven solutions play a crucial role in optimising the entire drug development lifecycle, from clinical trial preparation to execution. This not only reduces costs but also expedites the development of new treatments for patients.




OUR WHY FOR AFRICA
- Currently, 95% of cancer patients in Sub-Saharan Africa (SSA) are diagnosed too late.
- The population offers drug naïve patients in multiple disease areas that are accessible through careful planning and engagement.
- The ‘Rising Billion’ people in Africa are expected to reach 3-5 billion by 2050 representing half of the world’s population. The rising GDP and consumer power offers an attractive therapeutic market.
- Although the patients of African descent make up 17% of the world’s population (over 1Billion), Black People are underrepresented in clinical trials globally reported as less than 3% by the Genome-Wide Association Studies (GWAS).
OUR WHY FOR TECHNOLOGY
- Precision Data Collection: Harnessing AI to ensure precise and standardised data collection methods, reducing variability and enhancing the accuracy of clinical trial results.
- Real-time Monitoring and Insights for Improved Patient Engagement: Implementing AI-driven monitoring systems for real-time monitoring of patient data, enabling proactive intervention, and fostering active patient engagement, and addressing potential barriers, especially in remote or underserved areas of Africa.
- Automated Document Generation: Leveraging AI for automated document generation, to ensure that trial materials are accessible, consistent, and culturally sensitive, promoting inclusivity and ease of understanding for a diverse range of participants, reducing manual effort, and expediting the regulatory submission process.
- Enhanced Data Security and Compliance: Leveraging AI technologies for robust data security measures to build trust among patients, ensuring that their data is handled ethically and securely, thereby encouraging diverse participation in clinical trials.
- Efficient Data Analysis and Reporting: Employing AI algorithms for efficient data analysis, enabling faster identification of trends, potential issues, and insights that contribute to more informed decision-making and streamlined reporting processes.

Our Solutions